Does br2 react with alkenes
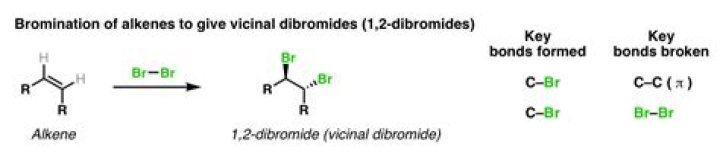
Description: Treatment of alkenes with bromine (Br2) gives vicinal dibromides (1,2-dibromides). Notes: The bromines add to opposite faces of the double bond (“anti addition”). Sometimes the solvent is mentioned in this reaction – a common solvent is carbon tetrachloride (CCl4).
What happens when Br2 reacts with an alkene?
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The double bond breaks, and a bromine atom becomes attached to each carbon. The bromine loses its original red-brown color to give a colorless liquid.
Do alkenes react quickly with bromine?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot. … This has the effect of ‘saturating’ the molecule, and will turn an alkene into an alkane.
Do alkanes react with Br2?
Alkanes are far less reactive than alkenes and will only react with bromine water in the presence of UV light. Under these conditions, alkanes undergo substitution reactions with halogens, and will slowly de-colourise bromine water.Does Br2 react with alkynes?
Bromine reacts rapidly with alkenes and alkynes. This can be used as a visual test to distinguish between alkanes, which do not react rapidly with bromine, and alkenes and alkynes.
Why bromine molecules react with the double bonds in alkenes?
That is, the electrons in the diatomic bromine molecule are repelled by the alkene and are pushed back along the molecule. The positively charged bromine atom acts as an electrophile, reacting with the double carbon bond. A pair of electrons from the carbon double bond move onto the positive bromine atom.
Is Br2 addition syn or anti?
Description: Treatment of alkenes with bromine (Br2) gives vicinal dibromides (1,2-dibromides). Notes: The bromines add to opposite faces of the double bond (“anti addition”).
How can you distinguish between alkynes and alkenes?
Alkenes and alkynes are hydrocarbon compound containing carbon atoms hydrogen atoms. Moreover, they are unsaturated compounds (have either double or triple bonds). The key difference between alkenes and alkynes is that the alkenes have carbon-carbon double bonds whereas the alkynes have carbon-carbon triple bonds.How alkenes and alkynes react with other substances?
Hydrohalogenation. Alkenes and alkynes can react with hydrogen halides like HCl and HBr. … If the alkene is asymmetric, the reaction will follow Markovnikov’s rule—the halide will be added to the carbon with more alkyl substituents.
How alkenes are converted to alkanes?To convert an alkene to an alkane, you must break the double bond by adding hydrogen to an alkene in the presence of a nickel catalyst, at a temperature of around 302 degrees Fahrenheit or 150 degrees Celsius, a process known as hydrogenation.
Article first time published onWhich type of hydrocarbons will not react with bromine?
Aromatic compounds are not very reactive. They will not react with Br2 or Cl2 under normal conditions. If the aromatic compound has an alkyl substituent, however, the alkyl group can be halogenated in the presence of UV light. The benzene ring will not react under these conditions.
How can you use Br2 as a test for the presence of an alkene in a product?
A simple test with bromine water can be used to tell the difference between an alkane and an alkene. An alkene will turn brown bromine water colourless as the bromine reacts with the carbon-carbon double bond. In fact this reaction will occur for unsaturated compounds containing carbon-carbon double bonds.
What do you mean by Hydrohalogenation of alkenes?
Reaction Overview: The hydrohalogenation of alkenes involves breaking a carbon to carbon double bond, followed by the electrophilic addition of a hydrogen atom and halogen. The halide will add to the more substituted carbon following Markovnikov’s rule. The product is a haloalkane also called an alkyl halide.
Where does Cl2 add to an alkene?
Reaction Overview: The alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as Cl2 or Br2 is added to a molecule after breaking the carbon to carbon double bond. The halides add to neighboring carbons from opposite faces of the molecule.
Does Lindlar's catalyst reduce alkenes?
Lindlar’s catalyst is a palladium catalyst poisoned with traces of lead and quinoline, that reduce its activity such that it can only reduce alkynes, not alkenes.
Are alkenes polar?
Alkenes are lighter than water and are insoluble in water due to their non-polar characteristics.
Which alkene gives a meso compound upon reaction with Br2 in ch2cl2?
trans-Hex-3-ene forms a meso compound when it reacts with bromine.
Which reagent follows syn addition with alkenes?
Complete step by step answer:The reagent giving syn addition are as follows: Osmium tetroxideOsO4followed by hydrolysis: it adds two –Oh groups at each carbon of the double bond in syn manner. Woodward reaction I2/AgOCOCH3(aq): cause hydroxylation in syn manner. Potassium permanganateKMnO4: diol addition in syn manner.
Why bromination of alkene is anti?
The result of this is the ring opening up with the two halogens on opposite sides as each other. This is anti stereochemistry, which is defined as the two bromine atoms come from opposite faces of the double bond. The product is that the bromines add on trans to each other.
Why do alkenes react with bromine water?
Bromine water is used in a simple test for unsaturated alkenes to distinguish them from saturated alkanes. The pi bond of the double bond opens up and two new carbon – bromine bonds (C–Br) are formed. This double bond makes alkenes much more reactive than alkanes, the bromine water test for alkenes is just one example.
What kind of reaction occurs when an alkene Decolourises bromine in tetrachloromethane?
The reaction with bromine happens at room temperature. If you have a gaseous alkene like ethene, you can bubble it through either pure liquid bromine or a solution of bromine in an organic solvent like tetrachloromethane. The reddish-brown bromine is decolourised as it reacts with the alkene.
Does toluene react with bromine?
Benzene and toluene do not react with bromine in the absence of a catalyst. Phenol is more reactive due to the presence of an electron donating hydroxyl group and reacts with bromine by a substitution reaction forming hydrogen bromide gas.
Which addition reaction is not undergo by alkenes?
Identify the addition reaction which is not undergone by the alkenes? Explanation: Alkenes do not undergo mercuration, indeed they undergo oxymercuration , a process in which an alkene is converted into an alcohol.
What type of reaction do alkynes undergo across the triple bond?
The principal reaction of the alkynes is addition across the triple bond to form alkanes. These addition reactions are analogous to those of the alkenes. Hydrogenation. Alkynes undergo catalytic hydrogenation with the same catalysts used in alkene hydrogenation: platinum, palladium, nickel, and rhodium.
Why do alkenes and alkynes undergo addition reactions?
Explanation: Alkenes and alkynes are unsaturated – they have π -bonds, so don’t have the full number of hydrogen that they could have. … The alkenes and alkynes want to form more σ -bonds and have a structure more like an alkane, so they undergo addition reactions.
Why are alkynes less stable than alkenes?
Its because electrons on multiple carbon-carbon bonds are more exposed and unstable. … The relative bond strength of a multiple carbon-carbon bonds such us alkyne and alkanes is smaller than normal single bond of an alkene thus making it less stable and reactive.
What makes alkanes differ from alkenes and alkynes?
The main difference between alkanes, alkenes, and alkynes is that alkanes form a single bond between carbon atoms while alkenes form a double bond while alkynes form a triple bond between the carbon atoms. Alkanes are also known as paraffin. The valencies of the four-carbon atom are filled by hydrogen atoms.
Are alkynes polar or nonpolar?
Alkynes are nonpolar, since they contain nothing but carbon and hydrogen, and so, like the alkanes and alkenes, they are not soluble in water, and are generally less dense than water.
What type of reaction is alkene to alkane?
An example of an alkene addition reaction is a process called hydrogenation.In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane.
What reaction forms alkenes?
Alkenes are generally prepared through β elimination reactions, in which two atoms on adjacent carbon atoms are removed, resulting in the formation of a double bond. Preparations include the dehydration of alcohols, the dehydrohalogenation of alkyl halides, and the dehalogenation of alkanes. Dehydration of alcohols.
What role do peroxides play in the reaction of HBr with alkenes select all that apply?
The correct answer to the given option is c. HBr H B r can be added to an alkene in the presence of peroxides (ROOR R O O R ). The function that the peroxide serve in this reaction is that it acts as a radical chain inhibitor.