What are normal hco3 levels
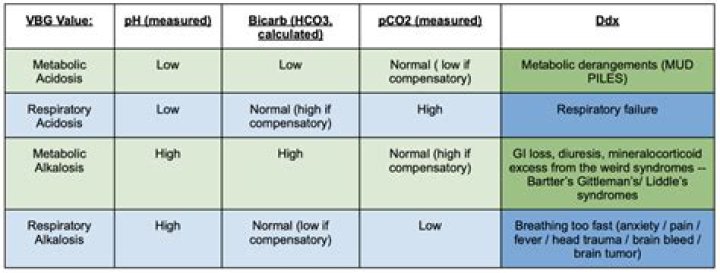
Arterial blood pH: 7.38 to 7.42. Oxygen saturation (SaO2): 94% to 100% Bicarbonate (HCO3): 22 to 28 milliequivalents per liter (mEq/L)
What does a high HCO3 level mean?
A high level of bicarbonate in your blood can be from metabolic alkalosis, a condition that causes a pH increase in tissue. Metabolic alkalosis can happen from a loss of acid from your body, such as through vomiting and dehydration.
Does HCO3 increase pH?
HCO3- level denotes metabolic/kidney effect. An elevated HCO3- is raising the pH and vice versa. If the pH is acidotic, look for the number that corresponds with a lower pH. If it is a respiratory acidosis, the CO2 should be high.
What do HCO3 levels indicate?
Low bicarbonate levels in the blood are a sign of metabolic acidosis. It is an alkali (also known as base), the opposite of acid, and can balance acid. It keeps our blood from becoming too acidic. Healthy kidneys help keep your bicarbonate levels in balance.What are signs of acidosis?
- rapid and shallow breathing.
- confusion.
- fatigue.
- headache.
- sleepiness.
- lack of appetite.
- jaundice.
- increased heart rate.
What is the difference between CO2 and HCO3?
Carbon dioxide (CO2) comes in a form of gas and is a waste product resulting from the body’s metabolism. … More than 90 percent of CO2 in the human blood is available as the bicarbonate (HCO3) form. The remaining carbon dioxide is either the dissolved form of the gas (CO2) or in carbonic acid form.
What causes low bicarbonate levels?
Examples of conditions that can cause a low bicarbonate level include: Addison disease. Chronic diarrhea. Diabetic ketoacidosis.
Which conditions can cause metabolic acidosis?
- Cancer.
- Carbon monoxide poisoning.
- Drinking too much alcohol.
- Exercising vigorously for a very long time.
- Liver failure.
- Low blood sugar (hypoglycemia)
- Medicines, such as salicylates, metformin, anti-retrovirals.
- MELAS (a very rare genetic mitochondrial disorder that affects energy production)
What happens when HCO3 is added to water?
Thus high HCO3 in water decreases the pH of water. HCO3 and pH are inversely proportional. When HCO3 increases , pH value decreases.
What blood pH is fatal?Normal arterial blood pH is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood pH below 7.35 is considered to be in acidosis (actually, “physiological acidosis,” because blood is not truly acidic until its pH drops below 7), and a continuous blood pH below 7.0 can be fatal.
Article first time published onWhat is Hyperchloremic acidosis?
Hyperchloremic acidosis is a disease state where acidosis (pH less than 7.35) develops with an increase in ionic chloride. Understanding the physiological pH buffering system is important. The major pH buffer system in the human body is the bicarbonate/carbon dioxide (HCO3/CO2) chemical equilibrium system.[1][2][3]
What is the most common cause of metabolic acidosis?
The most common causes of hyperchloremic metabolic acidosis are gastrointestinal bicarbonate loss, renal tubular acidosis, drugs-induced hyperkalemia, early renal failure and administration of acids.
What does a CO2 level of 31 mean?
Results are given in millimoles per liter (mmol/L) or milliequivalents per liter (mEq/L). Normal values in adults are 22 to 29 mmol/L or 22 to 29 mEq/L. Higher levels of carbon dioxide may mean you have: Metabolic alkalosis, or too much bicarbonate in your blood. Cushing disease.
How do you treat low bicarbonate levels?
Bicarbonate can balance out acid in your body. One way to increase bicarbonate levels is through alkali therapy, which could include taking sodium bicarbonate. Your doctor could also suggest taking another similar supplement, such as calcium citrate, calcium carbonate, or calcium acetate.
What is HCO3 in ABG?
HCO3- Concentration of hydrogen carbonate in. blood. Used to determine along with pH. and CO2 source of acid base imbalance.
Is TCO2 the same as HCO3?
Regulated primarily by the kidneys, HCO3 is the metabolic component of acid-base balance. … TCO2 is a measure of car bon dioxide which exists in several states: CO2 in physical solution or loosely bound to pro teins, HCO3 or CO3 ions, and carbonic acid (H2CO3).
Is HCO3 a strong or weak acid?
HCO3- (known as bicarbonate) is the conjugate base of H2CO3, a weak acid, and the conjugate acid of the carbonate ion. HCO3- acts as a base when mixed with a compound that is more acidic than itself (larger Ka) and as an acid when mixed with a compound that is more basic than itself (smaller Ka).
Is TCO2 the same as bicarb?
The reason why we use the term bicarb (HCO3-) instead of carbon dioxide (CO2) is because TCO2 is comprised of approximately 95% bicarb (HCO3-). … The rest of the total carbon dioxide (TCO2) includes dissolved CO2, carbonate ions and carbamino compounds.
Does H2CO3 lower pH?
Conclusion. The amount of carbon dioxide in water determines the pH of the water. The more CO2, the lower the pH, and vice versa.
Is metabolic acidosis serious?
Metabolic acidosis is a serious electrolyte disorder characterized by an imbalance in the body’s acid-base balance. Metabolic acidosis has three main root causes: increased acid production, loss of bicarbonate, and a reduced ability of the kidneys to excrete excess acids.
Can dehydration cause metabolic acidosis?
Metabolic acidosis develops when the body has too much acidic ions in the blood. Metabolic acidosis is caused by severe dehydration, drug overdoses, liver failure, carbon monoxide poisoning and other causes.
Can metabolic acidosis be reversed?
Metabolic acidosis can be reversed by treating the underlying condition or by replacing the bicarbonate. The decision to give bicarbonate should be based upon the pathophysiology of the specific acidosis, the clinical state of the patient, and the degree of acidosis.
Is apple cider vinegar acid or alkaline?
The pH of apple cider vinegar is about 2-3, which is considered mildly acidic. (pH is a measure of acidity, with 1 being the most acidic and 7 being neutral.) A substance known as ‘the mother’ (or vinegar mother) forms during the vinegar-making process.
What does a pH level of 7.4 mean?
Anything below 7.0 (ranging from 0.0 to 6.9) is acidic, and anything above 7.0 (from 7.1 to 14.0) is alkaline. The blood in your veins is slightly alkaline (pH = 7.4). The environment in your stomach is highly acidic (pH = 1 to 2).
How do you know if your body is alkaline or acidic?
A pH of 0 is totally acidic, while a pH of 14 is completely alkaline. A pH of 7 is neutral. Those levels vary throughout your body. Your blood is slightly alkaline, with a pH between 7.35 and 7.45.
How do you fix Hyperchloremic metabolic acidosis?
Correction of hyperchloremic acidosis is often accomplished with intravenous isotonic bicarbonate (150 mEq/L), which may require a substantial amount of volume.
When blood starts to become too acidic what kidneys respond by?
When there is too much acid in the body – acidosis – your kidneys try to excrete more acid into the urine and absorb more bicarbonate back into the body. When you have too little acid in your body, known as alkalosis, your kidneys try to excrete bicarbonate and conserve hydrogen ions.
What causes Hyperchloremic metabolic acidosis?
In general, the cause of a hyperchloremic metabolic acidosis is a loss of base, either a gastrointestinal loss or a renal loss.
What labs show metabolic acidosis?
The only definitive way to diagnose metabolic acidosis is by simultaneous measurement of serum electrolytes and arterial blood gases (ABGs), which shows pH and PaCO2 to be low; calculated HCO3- also is low.
Is co2 high or low in metabolic acidosis?
For a respiratory acidosis, the pCO2 is greater than 40 to 45 due to decreased ventilation. Metabolic acidosis is due to alterations in bicarbonate, so the pCO2 is less than 40 since it is not the cause of the primary acid-base disturbance.
What cancers cause low anion gap?
What does it mean if your Anion Gap result is too low? A low anion gap level is rare and indicates an abnormally high level of positively charged molecules. The most common cause of which, is multiple myeloma. Multiple myeloma is a cancer of a class of white blood cells called plasma cells.