What is a sintering process
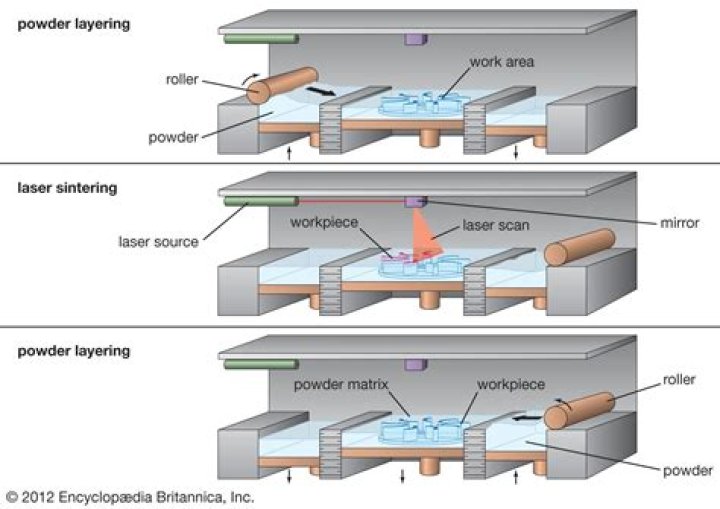
Sintering, which is also called ‘frittage,’ is the process of forming a solid mass of material through heat and pressure without melting to the point of liquefaction. … Sintering occurs naturally in mineral deposits, and is used as a manufacturing process for materials including ceramics, metals and plastics.
What is the purpose of sintering process?
Sintering is a heat treatment commonly used to increase the strength and structural integrity of a given material. Powder metallurgy processes use sintering to convert metal powders and other unique materials into end-use parts.
How do I stop sintering?
Catalyst sintering can be avoided by controlling the temperature of the burn front during the catalyst regeneration process. If the temperature gets too high, there can be localised sintering of the base, causing a loss of surface area.
What is a sintering in powder metallurgy and how it is done?
Sintering is a heat treatment applied to a powder compact in order to impart strength and integrity. The temperature used for sintering is below the melting point of the major constituent of the Powder Metallurgy material. … Reduction of the surface oxides from the powder particles in the compact.How do you make sinter?
Sintering is a thermal process (carried out at 1300 deg C to 1400 deg C) by which a mixture of iron ore, return fines, recycled products of the steel plant industry (such as mill scale, blast furnace dusts, etc.), slag forming elements, fluxes and coke fines are agglomerated in a sinter plant with the purpose of …
What are the benefits of sintering?
- Allows making complex geometries.
- Saving material.
- High precision.
- Stability in the process of big series.
- Good mechanical characteristics.
- Products ready for assembly.
- Cost economy compared with other processes.
What is sintering and its types?
Basically, sintering processes can be divided into three types: solid state sintering, liquid phase sintering and viscous sintering, which are all widely used in the industry. The driving force of sintering is the reduction in the total interfacial energy, which occurs via densification and grain growth.
Is sintering reversible?
Sintering is in general an irreversible process. Small catalyst particles have the highest possible relative surface area and high reaction temperature, both factors that generally increase the reactivity of a catalyst.What is sintering in dentistry?
Dental sintering furnaces are used to process dental zirconia after it has been milled into a crown, bridge, framework or other restoration. … The sintering process, which can involve pressure as well as heat, reduces the porosity and increases the density of ceramic materials such as zirconia.
Can sintering be reversed?[It can be completely reversed by reheating in an oxygen atmosphere or cooling slowly to room temperature from any temperature above 850 C].
Article first time published onHow are catalysts deactivated?
There are three fundamental reasons for catalyst deactivation, i.e. poisoning, coking or fouling and ageing. Poisoning can be reversible or irreversible, and with geometric or electronic effect. It can also be selective, nonselective and antiselective, depending on catalyst/poison affinity and kinetics.
What is sinter made up of?
Sintering is the process of fusing particles together into one solid mass by using a combination of pressure and heat without melting the materials. Common particles that are sintered together include metal, ceramic, plastic, and other various materials.
What is a sinter feed?
Sinter feed is an iron concentrate containing fine particles and moisture and, being similar to iron ore fines,”1 is at risk of liquefaction, resulting in cargo shift and loss of stability. Liquefaction is believed to have resulted in a number of casualties, causing the loss of many seafarers’ lives.
What do you mean by super Fluxed sinter?
Super flux sinters – These are the sinters where sufficient flux is added in the sinter mix for producing slags of desired basicity in blast furnace taking also into account the acidic oxides in the coke ash in addition to the other acidic oxides in the blast furnace burden.
How many types of sintering are there?
Basically, sintering processes can be divided into two types: solid state sintering and liquid phase sintering.
What is the powder metallurgy process?
Powder metallurgy is the process of blending fine powdered materials, pressing them into a desired shape or form (compacting), and then heating the compressed material in a controlled atmosphere to bond the material (sintering).
What is sintering with regards to a ceramic?
Sintering (Firing) of ceramic materials is the method involving consolidation of ceramic powder particles by heating the “green” compact part to a high temperature below the melting point, when the material of the separate particles difuse to the neghbouring powder particles.
What are the advantages and disadvantages of sintering process?
- Making of complex shapes directly, without secondary machining operations.
- Used for the shaping of materials which have a very high melting point.
- Reduce the porosity.
- Increase conductivity.
- Preservation of purity.
What is the dimensional accuracy in powder metallurgy?
What is the dimensional accuracy in powder metallurgy? Explanation: The powder metallurgy process yields net-shape, r near-net-shape parts, so that little or no machining is required to obtain a finished part in many cases.
What are the requirements for liquid phase sintering?
Therefore, for liquid-phase sintering, the requirements include that the liquid wets the solid particles, there is sufficient liquid present, and the solid is soluble in the liquid.
What is sintered zirconia?
The sintering process comprises a heating, a sintering, and a cooling phase (17). The sintering process may be altered in order to optimize the properties of zirconia. Even though, CAD-CAM technology has reduced the clinical operation times significantly, the zirconia sintering procedure still takes several hours.
What is sintering of zirconia?
During the production of high-tech ceramics, a ceramic powder is mixed with a binder and pressed to a green body. By thermal treatment the binder is removed (burned out) and the ceramic is sintered to the final part.
What advantages does Powder Metallurgy offer?
- Minimizes machining by producing parts at, or close to, final dimensions.
- Minimizes scrap losses by typically using more than 97% of the starting raw material in the finished part.
- Permits a wide variety of alloy systems.
- Produces good surface finish.
What is the difference between impregnation and infiltration?
Answer. Impregnation is when oil or other fluid is permeated into the pores of a sintered PM part. Infiltration is when a molten metal (other than the PM metal) is permeated into the pores of a sintered part.
What is pre sintering?
[prē′sint·ə·riŋ] (metallurgy) Heating a compact to a temperature lower than the final sintering temperature to facilitate handling or to remove a binder or lubricant.
Does sintering increase grain size?
This is then known as liquid-phase sintering (LPS) (Section 24.7). … After and during sintering, some grains will grow, consuming others. This process is known as grain growth and is essentially an Ostwald ripening process.
Does sintering decrease grain size?
Also, grains can diminish if there is a reaction during sintering, or a phase transition. But besides all, generally, observing one specimen, grain growth process can be regarded as irreversible. … In your system 1200oC is temperature relatively low for sintering of BaTiO3 and formed grains are small.
What is densification in material science?
An initial densification (decrease in void volume), the magnitude of which decreases as the void ratio (the ratio of void volume to the volume of the solid) approaches a minimum value. From: Handbook of Materials Behavior Models, 2001.
What is coke formation?
At low reaction temperatures, (<200°C) “coke” formation involves mainly condensation and rearrangement steps. … Their formation involves hydrogen transfer (acid catalysts) and dehydrogenation (bifunctional catalysts) steps in addition to condensation and rearrangement steps.
What is the deactivation?
1 : to make inactive or ineffective. 2 : to deprive of chemical activity deactivate an enzyme. Other Words from deactivate. deactivation \ -ˌak-tə-ˈvā-shən \ noun.
What are poisons in catalysis?
catalyst poison, substance that reduces the effectiveness of a catalyst in a chemical reaction. … In practice, however, poisons, which come from the reacting substances or products of the reaction itself, accumulate on the surface of solid catalysts and cause their effectiveness to decrease.