What is mass defect example
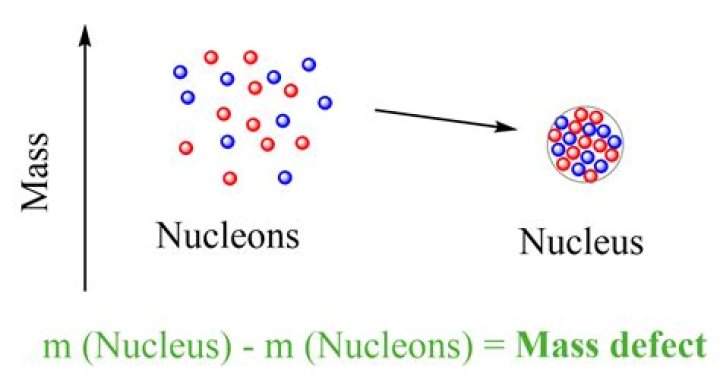
The actual mass of the atomic nucleus is always less than the mass of protons and neutrons present in the nucleus. When a nucleus is formed, energy is released. This missing mass is known as the ‘mass defect’ and it accounts for the energy released. …
Why does mass defect exist?
Mass defect is caused due to nuclear binding energy. This energy is solely responsible for keeping the nucleuons together. This is done through an exchange of virtual pions. Their mass is determined using the uncertainty principle.
How is mass defect determined?
To calculate the mass defect: add up the masses of each proton and of each neutron that make up the nucleus, subtract the actual mass of the nucleus from the combined mass of the components to obtain the mass defect.
What is the mass defect of an atom?
Careful measurements have shown that the mass of a particular atom is always slightly less than the sum of the masses of the individual neutrons, protons, and electrons of which the atom consists. The difference between the mass of the atom and the sum of the masses of its parts is called the mass defect (Δm).Is mass defect positive or negative?
Nuclear mass defect is a negative value and has the same sign for all elements and therefore binding energies as the energy that keeps the nucleus together will all have the same sign as expected.
What is mass defect topper?
Mass defect is the amount by which the mass of an atomic nucleus differs from the sum of the masses of its constituent particles, being the mass equivalent of the energy released in the formation of the nucleus. Nuclear binding energy per nucleon for the given atom=ABE
What is the difference between mass excess and mass defect?
(Hydrogen-1 nucleus being exception which has only one proton). the combined mass is less than sum of individual masses. This is known as mass defect or sometimes also called mass excess. It represents the energy that was released when the nucleus was formed, called binding energy of nucleus.
How is mass defect related to binding energy?
The mass defect of a nucleus is the difference between the total mass of a nucleus and the sum of the masses of all its constituent nucleons. The binding energy (BE) of a nucleus is equal to the amount of energy released in forming the nucleus, or the mass defect multiplied by the speed of light squared.Who discovered mass defect?
The mass defect and binding energy are related by Albert Einstein’s formula, E = mc2. In 1905, Einstein developed the special theory of relativity. One of the implications of this theory was that matter and energy are interchangeable with one another.
Which is magic number?The seven most widely recognized magic numbers as of 2019 are 2, 8, 20, 28, 50, 82, and 126 (sequence A018226 in the OEIS). For protons, this corresponds to the elements helium, oxygen, calcium, nickel, tin, lead and the hypothetical unbihexium, although 126 is so far only known to be a magic number for neutrons.
Article first time published onWhat is mass defect of a nucleus express it mathematically?
Mass defect is the difference in the mass of nucleus and its constituents(neutrons and protons). It is denoted by ΔM. Mathematically :- ΔM = [Z mp+ (A-Z) mn]- M.
What is mass defect how is it related to stability of the nucleus?
Answer: Because the magnitude of the mass defect is proportional to the nuclear binding energy, both values indicate the stability of the nucleus. Just as a molecule is more stable (lower in energy) than its isolated atoms, a nucleus is more stable than its isolated components.
What is responsible for the mass defect when protons and neutrons combine to form a nucleus?
The mass difference comes from an extra neutron or proton. b. The mass defect comes from the electrons of the newly formed atom.
What are magic nuclei?
Magic nuclei are those having the precise number of protons or neutrons required to fill a spherical set of related orbitals called a ‘shell’. Nuclei with magic neutron or proton numbers are characterized by a stronger binding, greater stability, and, therefore, are more abundant in nature.
What is the mass defect in grams of Cu 63?
A copper-63 nucleus has a mass defect of 0.59223 amu.
What is the proton symbol?
ParticleSymbolChargeelectrone–1protonp++1neutronno0
What element has the largest mass defect?
Sn has the largest mass defect, Δm per nucleon since it has the highest binding energy, E.
Why is mass defect important for nuclear reactions?
Mass Defect is the amount of matter converted to energy on formation of an atomic nucleus. Cause is to achieve a more stable nuclear configuration.
What are the magic numbers in nuclear physics?
The magic numbers for nuclei are 2, 8, 20, 28, 50, 82, and 126. Thus, tin (atomic number 50), with 50 protons in its nucleus, has 10 stable isotopes, whereas indium (atomic number 49) and antimony (atomic number 51) have only 2 stable isotopes apiece.
What is mass defect at ground state mass of a nucleus is always less than the sum of mass of its constituents protons and neutrons give reason?
Since the nucleus is made up of neutron and proton so it is expected that the mass of the nucleus is equal to the total mass of its individual constituents however, it found less than its total mass of its constituents because during the formation of the nucleus some amount of energy gets released.
Why is 3 a sacred number?
Throughout human history, the number 3 has always had a unique significance, but why? The ancient Greek philosopher, Pythagoras, postulated that the meaning behind numbers was deeply significant. In their eyes the number 3 was considered as the perfect number, the number of harmony, wisdom and understanding.
Is 7 a magic number?
Examining a new model detailing the recall of information within the human brain. This limit, which psychologists dubbed the “magical number seven” when they discovered it in the 1950s, is the typical capacity of what’s called the brain’s working memory. …
Why is 114 not a magic number experimentally?
Super-heavy elements like 114 usually only exist for fractions of a second. Scientists make these elements one atom at at time by smashing together the nuclei of lighter elements. … Scientists hypothesized that element 114 had that magic number, a hypothesis this experiment has disproved.
What does mean by mass defect establish relation between mass defect and nuclear binding energy and hence write the expression for binding energy per nucleon?
The energy equivalent to mass defect is used in binding the nucleons and is called the binding energy. If Δm is the mass defect of a nucleus, then according to Einstein’s mass energy relation, Binding energy = Δmc2( in joule ). Binding energy = ([Zmp+(A−Z)mn]−mN(ZXA))c2( in joule ).
How does mass defect relate to binding energy in the nucleus apex?
How does mass defect relate to nuclear binding? Mass defect is the difference between the mass of an atom and the sum of the masses of its particles. The nuclear binding energy is the energy released when a nucleus is formed from nucleons. … more binding energy per nucleon creates a more stable nucleus.