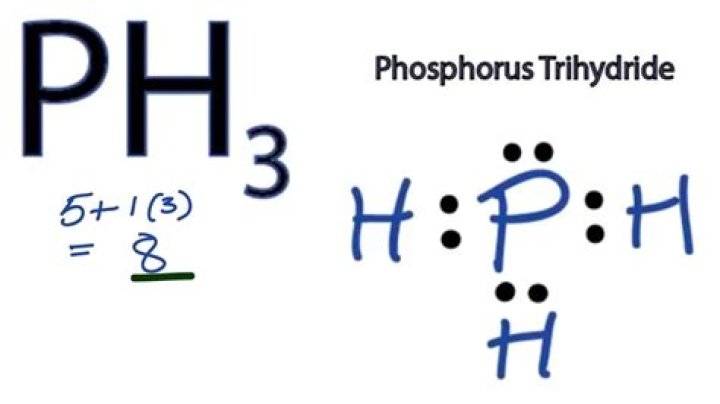
What is ph3 used for
Phosphine (PH3), a highly toxic, flammable gas, is used in the semiconductor industry, in chemical processing, and more commonly in fumigation of grain, food products, and tobacco, prior to international transport.
What are the uses of PH3?
- In semiconductor industries, it is used in small amounts as a dopant.
- PH3 is used in Holme’s signal due to its property of spontaneous combustion.
What is PH3 called?
phosphine (PH3), also called hydrogen phosphide, a colourless, flammable, extremely toxic gas with a disagreeable garliclike odour. Phosphine is formed by the action of a strong base or hot water on white phosphorus or by the reaction of water with calcium phosphide (Ca3P2).
How do you get PH3?
Laboratory preparation: It is usually obtained by boiling white phosphorus with 30-40% solution of caustic soda in an inert atmosphere of CO2. Phosphine so obtained is impure. It is passed into an aqueous solution of hydrogen iodide, PH4I is formed. PH4I is heated with KOH or NaOH, pure phosphine is obtained.What is a phosphine detector?
A phosphine detector (PH3 detector) is used to measure concentrations of this gas in the air and to trigger an alert when the level becomes dangerous. … Phosphine is a colorless, extremely toxic gas, identifiable by its fishy odor. It is also very explosive and can ignite spontaneously in the air.
Is PH3 molecular or ionic?
Phosphene PH3 is a covalent compound. The P-H bond has more covalent character. This is because the bonds are formed as a result of overlapping between 3p orbital (of phosphorus) and 1s orbital (of hydrogen).
Does PH3 react with water?
NamesMelting point−132.8 °C (−207.0 °F; 140.3 K)Boiling point−87.7 °C (−125.9 °F; 185.5 K)Solubility in water31.2 mg/100 ml (17 °C)
What does PH3 fumes in moist air?
In the presence of moisture, the hydrolysis of PCl3 gives fumes of phosphorus acid and hydrochloric acid.What is the charge of PH3?
Property NameProperty ValueReferenceFormal Charge0Computed by PubChemComplexity0Computed by Cactvs 3.4.8.18 (PubChem release 2021.05.07)
What is the oxidation state of PH3?Thus in PH3, the H is in oxidation state of +1 and P is in oxidation state -3.
Article first time published onIs PH3 an acid or base?
PH3 is a weaker base than NH3 because the lone pair within NH3 has a higher energy and thus serve better as a better base. Each subsequent fall on the periodic table has less energy and thus becomes less basic which in turn means it is more acidic.
What is the metal of PH3?
It’s a non metal / non metal and is molecular. Also known as phosphine. The IUPAC name for this compound is phosphane. You’ll sometimes see this called phosphorus trihydride, but that is a less common name.
What type of metal is PH3?
Phosphine, also known as [PH3] or fosfano, is a member of the class of compounds known as homogeneous other non-metal compounds. These compounds are inorganic nonmetals in which the largest atom belongs to the class of ‘other nonmetals’.
Is PH3 a gas?
Phosphine (PH3), a highly toxic, flammable gas, is used in the semiconductor industry, in chemical processing, and more commonly in fumigation of grain, food products, and tobacco, prior to international transport.
What intermolecular forces are present in PH3?
The intermolecular forces between phosphine(PH3) molecules are dipole- dipole forces/Van der Waals forces, whereas the intermolecular forces between ammonia(NH3) molecules are hydrogen bonds.
Why is PH3 a gas?
NH3 is a liquid at room temperature while PH3 is a gas. This is because in NH3 the molecules are associated to each other through intermolecular hydrogen bonds whereas in PH3 there is no such type of association between the molecules, hence, it exist as gas.
What is found when PH3 reacts with an acid?
Answer: Phosphine react with nitric acid to produce phosphoric acid, nitrogen dioxide and water.
Is PH3 a neutral ligand?
The compound phosphine (PH3) is extremely important in coordination chemistry due to its large number of derivatives which can be used as L-type ligands (2 electron donor neutral ligands) for many metal complexes.
Does PH3 have hydrogen bonding?
The molecule PH3 does not have hydrogen bonding as it does not qualify for hydrogen bonding since the hydrogens are not attached to either fluorine,…
Is NH3 or PH3 more ionic?
Which one is more covalent, NH3 or PH3? And why? – Quora. The electronegativity of P and H is almost same so there is no electronegativity difference. And more the electronegativity difference more it has ionic character so PH3 will be more covalent.
Is PH3 polar?
PH3 is a polar molecule because it has a bent structure due to lone pairs of electrons and electron-electron repulsion. Phosphorus’s electronegativity is a nonpolar molecule because it is the same, but since Phosphorus has a lone pair, PH3 is a polar molecule.
In what way can it be proved that PH3 is basic in nature?
PH3 reacts with acids like HI to form PH4I. This shows that PH3 is basic in nature. Due to the presence of a lone pair of electrons on P,PH3 acts as a Lewis base in the above reaction.
Why does PCl3 fume in Monster?
In the presence of moisture, PCl3 undergoes hydrolysis to release fumes of HCl gas. … In the presence of moisture, the hydrolysis of PCl3 gives fumes of phosphorus acid and hydrochloric acid.
Is phosphorus an acid?
phosphorous acid (H3PO3), also called orthophosphorous acid, one of several oxygen acids of phosphorus, used as reducing agent in chemical analysis. It is a colourless or yellowish crystalline substance (melting point about 73° C, or 163° F) with a garliclike taste.
What happens when H3PO3 is heated?
On heating, H3PO3 decomposes into phosphoric acid and phosphine.
What does high oxidation mean?
While some new oils may produce an oxidation value due to the presence of synthetic esters, increases in oxidation generally represent degradation of the fluid. In situations with extremely high temperatures, even localized hot spots, both oxidation and nitration will increase. …
What is the oxidation number of NaH2PO4?
Thus , oxidation number of P in NaH2PO4 is + 5 .
Can PH3 act as lewis acid?
Answer: d)PH3 is a base not a lewis acid!
Why is PH3 act as Lewis base?
PH3 reacts with acids like HI to form PH4I which shows that it is basic in nature. Due to lone pair on phosphorus atom, PH3 is acting as a Lewis base in the above reaction.
Why is PH3 not a lewis acid?
A Lewis base is an electron-pair donor. Only PH3 has a pair of nonbonding electrons and can act as a donor.
What is Holmes signal?
Answer: Holmes signal – A mixture of calcium carbide and calcium phosphide placed in a container which reacts with water to produce gases (PH3 and C2H2) which burn and thus guide the ships in sea.so PH3 gas that is called PHOSPHINE is used in holmes signal.