What is the boiling of H2S
Hydrogen sulfide appears as a colorless gas having a strong odor of rotten eggs. Boiling point -60.2°C.
Why is H2S boiling point low?
Water is more polar than hydrogen sulfide because the oxygen atom is more electronegative than the sulfur atom. Also, hydrogen sulfide has weaker intermolecular forces (dipole forces, vander waal’s forces) compared to water resulting in a lower melting point and boiling point.
Why is H2O stronger than H2S?
The H–F bond is more polar than H–O so is easier to break HF into H+ and F- than to break H2O into H+ and OH-. … S is much larger atom than O, so the H–S bond is much longer and weaker than H–O; hence H2O is weaker acid than H2S.
Why H2S has high boiling point?
This is an account of intermolecular hydrogen bonding which is present in the molecules of H2O but is absent in the molecules of H2S. As a result, both melting and boiling point of water are higher than those of hydrogen sulphide.Does H2S have higher boiling point?
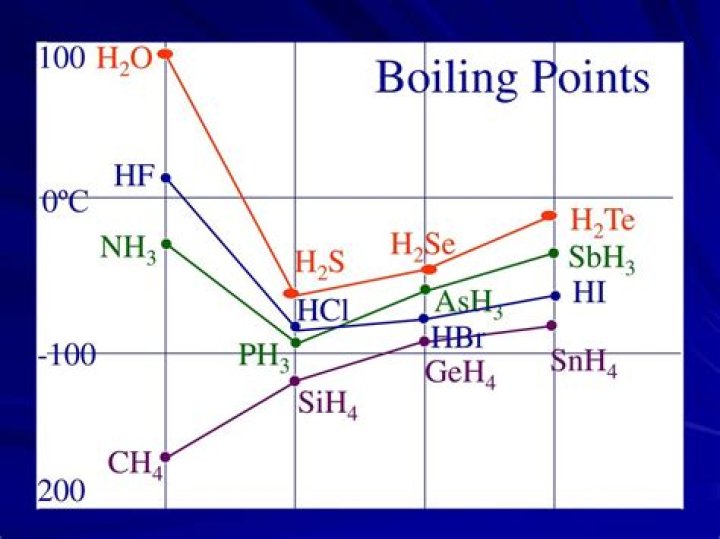
H2S has the lowest boiling point. As the molar mass increases boiling point of the hydrides increases except H2O. The exception of water is due to intermolecular hydrogen bonding among the water molecules. Water molecules are in associated state.
Why H2S has higher boiling point than HCl?
Both HCl and H2S have 18 electrons, BUT the area over which they are spread in H2S is greater than for HCl. Therefore, we can conclude that H2S has greater London dispersion forces which contribute to the higher boiling point.
Why does H2S have lower boiling point than H2S?
This essentially means H2Se is a bigger molecule than H2S meaning the Van Der Waal forces (London forces) on H2Se is stronger than on H2S which means it takes more energy to break the intermolecular forces for H2Se than H2S– this leads to H2S having the lower boiling point.
Is H2S gas explosive?
Hydrogen sulfide is a highly flammable, explosive gas, and can cause possible life-threatening situations if not properly handled. In addition, hydrogen sulfide gas burns and produces other toxic vapors and gases, such as sulfur dioxide.Is H2S more polar than H2O?
O is more electronegative than S,this is the reason why H2O is more polar than H2S.
Why is H2S called sulfide?What is Hydrogen Sulfide? Hydrogen sulfide is a chemical compound with the chemical formula H2S. This means it is made of two hydrogen atoms, and one sulfide.
Article first time published onDoes H2S or NH3 have a higher boiling point?
NH3 SO2 N2 H2S H2O. Therefore, H2O has the highest boiling point.
Is HS stronger than H2S?
The greater the ability of a species to accept a H+ from another species, the greater its base strength. Organic chemists customarily compare the strength of bases using the strengths of their conjugate acids, measured as pKa. HF is a stronger acid than H2S. … Thus, HS ¯ is a stronger base than F ¯.
Which is stronger H2S or HCl?
HCl is a stronger acid than H2S because Cl is more electronegative than S. b.) HClO4 is a stronger acid than HClO2 because it has more oxygens around its central atom.
Is H2Se or H2S a stronger acid?
In binary acids such as H2S and H2Se, the H–Se bonds is longer than the H–S bonds as Se is larger than S. The H–Se bond is therefore weaker than the H–S bond and H2Se is thus a stronger acid than H2S.
Does H2S have stronger intermolecular forces?
H2S has dipole-dipole, so it is stronger than I2. H2O has hydrogen bonding, so it is stronger than H2S. These compounds are all the same shape, and are all non-polar. Therefore, the difference in London dispersion forces are more important for these compounds.
Which has a higher boiling point HI or HF?
The confusion can occur why HF has a higher boiling point than the HI. The reason is the presence of strong hydrogen bonding in the HF, as hydrogen intermolecular interactions are greater than the van der Waals forces. So, HF has a higher boiling point than the HI.
Why does h20 have a high boiling point than H2Se?
H2O molecules undergo hydrogen bonding. … On the other hand, H2Se and H2Te contain hydrogens that are not attached to electronegative elements, so they cannot undergo hydrogen bonding, and they possess boiling points that are significantly lower than water.
Why is H2S less polar than water?
This means that hydrogen sulfide is not as polar as water. … By comparison, the smaller dipole moment of a hydrogen sulfide means a less significant separation of charge between the partial-positive end of the molecule and the partial negative end of the molecule → hydrogen sulfide is not as polar as water.
Why is H2S more polar than HCl?
HCl has more ionic bonds than H2S because there is a greater difference of electronegativity between the hydrogen and chlorine atoms than between the hydrogen and sulfur atoms. Additionally, because of this, the HCl molecule is also more polar than the H2S molecule.
What is the boiling point of h2o?
The boiling point of a liquid varies according to the applied pressure; the normal boiling point is the temperature at which the vapour pressure is equal to the standard sea-level atmospheric pressure (760 mm [29.92 inches] of mercury). At sea level, water boils at 100° C (212° F).
What type of intermolecular forces does H2S have?
Dipole – dipole forces – Intermolecular force exhibited by polar molecules in which positive end of one dipole attracts the negative end of another polar molecule. e.g. HBr & H2S.
Is H2S polar or nonpolar?
Name of moleculeHydrogen sulfidePolarityPolar natureStructureBent shapeNo of lone pairsTwoValence electronsEight
Why H2S is less acidic than H2Te?
H2Te has less bond dissociation enthalpy than H2S . So, less energy is required to break H2Te bond & releasing [H]+ is easier & hence the acidity of H2Te is higher. Whereas, H2S has High Bond Dissociation Energy & hence the acidity of H2S is less.
What ppm is H2S flammable?
Very high levels can cause unconsciousness and even death. DANGEROUS FIRE HAZARD. OSHA: The legal airborne permissible exposure limit (PEL) is 20 ppm not to be exceeded at any time, and 50 ppm as a maximum peak, not to be exceeded during any 10-minute work period.
At what ppm is H2S explosive?
= Immediately Dangerous to Life and Health. S.T.E.L. H2S = 15 ppm Page 17 Exposure Limits Knocked out immediately, death in 35 minutes.
What causes H2S to explode?
Heat from fire can cause a rapid build-up of pressure inside cylinders. Explosive rupture and a sudden release of large amounts of gas may result.
What happens when h2s burns in air?
Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
Why is it h2s and not sh2?
NamesChemical formulaH2SMolar mass34.08 g·mol−1AppearanceColorless gasOdorPungent, like that of rotten eggs
Is h2s ionic or covalent?
Name of MoleculeHydrogen sulfideChemical formulaH2SMolar mass34.08 g/molBond typeCovalentNatureAcid
Why does h20 have a higher boiling point than HF and NH3?
H2O has a higher boiling point than NH3 because (i) the H-bonds are stronger and (ii) it contains twice as many H-bonds. H2O has a higher boiling point than HF because it contains twice as many H- bonds, despite these being individually weaker.
Is H2S analogous to ammonia?
So, when there are two elements close to each other on the periodic table, they tend to have similar attributes. For instance, water (H2O), ammonia (NH3), and hydrogen sulfide (H2S) should be very similar molecules.