What occurs in sublimation
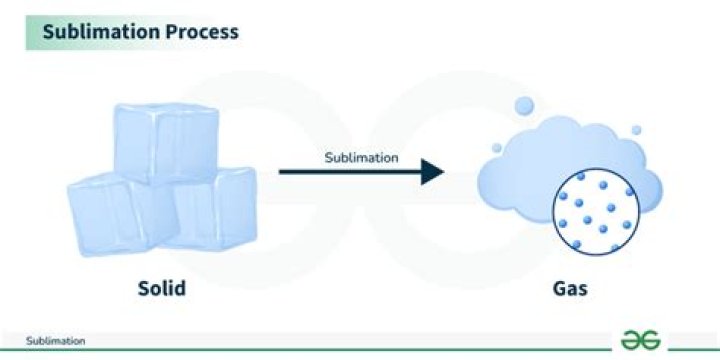
Sublimation is the conversion between the solid and the gaseous phases of matter, with no intermediate liquid stage. For those of us interested in the water cycle, sublimation is most often used to describe the process of snow and ice changing into water vapor in the air without first melting into water.
What happens during sublimation chemistry?
Sublimation: The process in which a solid transforms into a gas phase without first melting to form a liquid phase. … Dry ice is solid carbon dioxide at -78 oC. This substance converts to the gas phase by sublimation without first forming liquid carbon dioxide.
What happens to heat during sublimation?
In sublimation, a solid substance is volatilized by heating and the vapor is condensed back to the solid at a cooled surface.
Which phase occurs during sublimation?
Sublimation is the process of transformation directly from the solid phase to the gaseous phase, without passing through an intermediate liquid phase.How does sublimation process occur?
Sublimation is caused by the absorption of heat which provides enough energy for some molecules to overcome the attractive forces of their neighbors and escape into the vapor phase. Since the process requires additional energy, it is an endothermic change.
What is sublimate in chemistry?
Sublimate is defined as to cause the change of a gas into a solid or a solid into a gas without becoming a liquid, or to have a refining effect on someone or something. An example of sublimate is dry ice turning directly into carbon dioxide from solid form. … (chemistry) A product obtained by sublimation.
What is the process of sublimation?
The sublimation printing process Well, sublimation printing uses heat to essentially bring ink and fabric together as one. First, a design is printed onto special paper. The inks that are used turn into gas when brought under heat, then combine with the fabric and permanently print onto the fabric.
What are 3 examples of sublimation?
- Dry Ice. As mentioned earlier, dry ice is one of the most popular examples of sublimation in real life. …
- Water. …
- Specialized Printers. …
- Moth Balls. …
- Freeze Drying. …
- Air Fresheners.
Which substances undergo sublimation process?
Camphor, maphthalene, ammonium chloride, iodine, and dry ice are some substances which undergo sublimation.
What is the deposition phase?Deposition is the phase transition in which gas transforms into solid without passing through the liquid phase. … The reverse of deposition is sublimation and hence sometimes deposition is called desublimation.
Article first time published onWhat is fusion in phase changes?
Fusion. Fusion occurs when a substance changes from a solid to a liquid. Prior to melting, strong intermolecular bonds or attractions hold the atoms, molecules or ions that comprise a solid substance tightly together in the solid form.
What are the 5 phase changes?
Phase Change: Evaporation, Condensation, Freezing, Melting, Sublimation & Deposition.
Is energy released or absorbed during sublimation?
Sublimation, a substance going straight from solid to gas phase, also involves the absorbing of energy.
What energy is involved in sublimation?
The energy associated with a transition from solid directly to gas is called the latent heat of sublimation. Sublimation also happens with snow. This means that when the air is particular dry (low humidity) the water turns directly from snow or ice into water vapour without being liquid at all.
What factors affect sublimation?
Sublimation fastness depends on many factors, such as (i) temperature of exposure, (ii) time of exposure, (iii) shade depth, (iv) chemical structure of the fibre and (v) chemical structure of the dye (Shakra and Ali, 1992).
What would happen if sublimation didn't occur?
In the winter, you would expect that ice would form on the clothes, but that didn’t happen. Even in freezing weather, the water might freeze initially, but would eventually go off as a vapor – from solid ice directly to the gas stage.
What are examples of deposition?
Examples. One example of deposition is the process by which, in sub-freezing air, water vapour changes directly to ice without first becoming a liquid. This is how frost and hoar frost form on the ground or other surfaces. Another example is when frost forms on a leaf.
What is sublimation process give an example to define it?
Sublimation is the process by which a substance changes from solid state directly to vapour state. Example : dry ice, naphthalene balls etc.
What is sublimation and why does it occur?
The process in which a solid changes directly to a gas is called sublimation. It occurs when the particles of a solid absorb enough energy to completely overcome the force of attraction between them. … Solid carbon dioxide changes directly to the gaseous state.
What is sublimation in chemistry class 6?
Sublimation is the process in which a solid changes directly to a gas without going through the liquid state. Solid carbon dioxide is an example of a substance that undergoes sublimation.
What is sublimation in analytical chemistry?
Sublimation is a purification technique for solids and in the context of this book, for organic compounds with lower melting points. Sublimation describes the process of a solid becoming a gas, without passing through the liquid state. … The organic compound will start to sublime forming a gas.
Why does naphthalene undergo sublimation?
Sublimation means when a solid changes directly into gaseous state without going into the liquid state. Naphthalene undergoes sublimation because naphthalene is highly volatile in nature and there is weak force of attraction between its molecules. As a result, it undergoes sublimation even at room temperature.
What are sublimation 5 examples of sublimation?
- Dry ice. Carbon dioxide (CO 2 ) can be liquefied first and then frozen, to make dry ice. …
- Polar evaporation. …
- Snow in the mountains. …
- The disappearance of naphthalene. …
- Arsenic treatment. …
- Iodine treatment. …
- Frost formation. …
- Planetary accretion.
What are examples of sublimation and deposition?
This change from a solid to a gas is called sublimation. The reverse process of a gas going to a solid is known as deposition. As an example, solid carbon dioxide (dry ice) will sublimate to produce gaseous carbon dioxide at room temperature. Evaporation is the process by which a liquid transitions to a gas.
What are Sublimable solids?
-Sublimable solid – The solid compounds which undergo sublimation are called sublimable solid. -Non – sublimable – Those solid compounds which directly on heating without passing through the liquid phase are called non – sublimable compounds. -Iodine is a sublimable solid.
How does sublimation and deposition happen?
Sublimation is the process by which molecules go directly from solid into the vapor or gas phase. Deposition is the process by which molecules go directly from the gas phase into the solid phase. Deposition chemistry occurs when molecules settle out of the gas phase and into the solid phase.
What happens during deposition?
At a deposition, a person appears at a specified time and place and gives sworn testimony—under oath, usually with a court reporter present so that a record is made. Depositions typically occur during the discovery phase of a personal injury case (after the filing of a lawsuit, but before trial or settlement).
How does gas deposition occur?
The opposite of sublimation is deposition. This is the process in which a gas changes directly to a solid without going through the liquid state. It occurs when gas particles become very cold.
What is vaporization phase change?
vaporization, conversion of a substance from the liquid or solid phase into the gaseous (vapour) phase. If conditions allow the formation of vapour bubbles within a liquid, the vaporization process is called boiling.
Is gas to liquid Fusion?
Condensation is when a gas is changed into a liquid.
What happens to energy during a phase change?
The energy that is changing during a phase change is potential energy. During a phase change, the heat added (PE increases) or released (PE decreases) will allow the molecules to move apart or come together. Heat absorbed causes the molecules to move farther apart by overcoming the intermolecular forces of attraction.