Why is copper iodide white
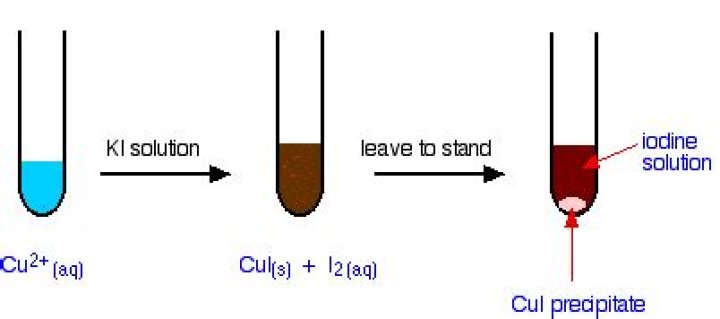
The copper(I) iodide is virtually insoluble in water, and so the disproportionation reaction doesn’t happen. Similarly copper(I) chloride can be produced as a white precipitate (reaction described below). Provided this is separated from the solution and dried as quickly as possible, it remains white.
What Colour is CuI?
NamesChemical formulaCuIMolar mass190.45 g/molAppearanceWhite to tan colored powderOdorodorless
Is CuI aqueous?
Preparation. In the laboratory, copper(I) Iodide is prepared by simply mixing an aqueous solutions of sodium or potassium iodide and a soluble copper(II) salt such copper sulfate. … CuI is poorly soluble in water (0.00042 g/L at 25 °C), but it dissolves in the presence of NaI or KI to give the linear anion [CuI2]−.
What color is copper II iodide?
Compound FormulaICuMolecular Weight190.45AppearanceLight Brown PowderMelting Point606° C (1,123° F)Boiling Point1,290° C (2,354° F)Why is CuI Colourless?
One: CuX (X = Cl, Br and I) and all colourless powders because they have no electronic transitions with energies in the visible spectrum.
Does Cu2I2 exist?
Cu2I2 does not exist under normal circumstances, although it is conceivable it could exist in a transition state, say in a molecular beam, where 2 CuI molecules collide to form said Cu2I2 dimer molecule.
Is CuI paramagnetic or diamagnetic?
In Cu+ the electronic configuration is 3d10 completely filled d- shell thus it is diamagnetic. thus it has one unpaired electron in d- subshell thus it is paramagnetic.
Is CuI ionic or covalent?
CuI is an ionic compound that has each molecule made from one atom of copper (Cu) and one atom of iodine (I). The copper atom is positively charged and the iodine is negatively charged, so there is an ionic bond between them.How do you purify CuI?
The crude CuI formed may be purified by dissolving in hot concentrated KI solution and recrystallise upon cooling and dilution. UV-vis spectroscopy can be used to determine the purity of the sample obtained.
What is Colour of cucl2?Copper chloride appears as a yellowish-brown powder (the anhydrous form) or a green crystalline solid (the dihydrate).
Article first time published onWhat is CuI in chemistry?
Copper(I) iodide.
What is Cui specified?
CUI Specified – The subset of CUI for which the authorizing law, regulation or Government- wide policy contains specific handling controls that it requires or permits agencies to use that differ from those for CUI Basic.
Why is CuI2 not stable?
Iodide ions are strong reducing agents. Therefore, Copper (II) Iodide reduces to insoluble copper (I) iodide. Thus making CuI2, CuI. … It isn’t stable because of electrochem and the solubility.
Is iron iodide soluble?
NamesMelting point587 °C (1,089 °F; 860 K)Boiling point827 °C (1,521 °F; 1,100 K)Solubility in watersolubleMagnetic susceptibility (χ)+13,600·10−6 cm3/mol
Why cupric salts are blue?
That’s what happens with the metal. In compounds copper sulphate, the blue colour is due to the light energy being used to promote or excite electrons that are in the atom of the copper when it’s combined with other things such as the sulphate or carbonate ions and so on.
Why is CU II blue?
Copper(II) ions in solution absorb light in the red region of the spectrum. The light which passes through the solution and out the other side will have all the colors in it except for the red. We see this mixture of wavelengths as pale blue (cyan).
Why cu1 is Colourless and cu2 is blue in colour?
Cu+ has completely filled orbital: [Noble gas] 3d10, where as Cu2+ has partially filled orbital : [Noble gas]3d9. Hence Cu+ is colourless but Cu2+ is coloured.
Is NI diamagnetic?
d) Ni(CO)4 is diamagnetic; [Ni(CN)4]2- and NiCl42- are paramagnetic. * The valence shell electronic configuration of ground state Ni atom is 3d8 4s2. * All of these 10 electrons are pushed into 3d orbitals and get paired up when strong field CO ligands approach Ni atom. … Thus Ni(CO)4 is diamagnetic.
What Colour is Cu+?
Cu+ has completely filled orbital: [Noble gas] 3d10, where as Cu2+ has partially filled orbital : [Noble gas]3d9. Hence Cu+ is colourless but Cu2+ is coloured.
Is O2 diamagnetic in nature?
When the electrons are paired in any orbital their net Spin is zero and electrons are called diamagnetic electrons. … This theory proves that an oxygen molecule has two unpaired electrons and is paramagnetic in nature.
What is the correct name for al2o3?
PubChem CID9989226Molecular FormulaAl2O3SynonymsALUMINUM OXIDE Aluminium oxide 1344-28-1 gamma-Alumina dialuminum;oxygen(2-) More…Molecular Weight101.961Component CompoundsCID 190217 (Oxide) CID 5359268 (Aluminum)
Why is CuO black?
Explanations : Heated copper metal reacts with oxygen to form the black copper oxide. The copper oxide can then react with the hydrogen gas to form the copper metal and water. When the funnel is removed from the hydrogen stream, the copper was still be warm enough to be oxidized by the air again.
Does iron iodide exist?
Compounds with iodine in formal oxidation state −1 are called iodides. This can include ionic compounds such as caesium iodide or covalent compounds such as carbon tetraiodide. … Iron(III) iodide does not exist because iron(III) ions oxidize iodide ions in aqueous solution.
Does fei3 exist?
FeI3 does not exist.
Which halide does not exist?
Thus, all the three halogens except iodine react with copper and form their consecutive halides but $Cu{I_2}$ does not exist.
Is Iodine an ion?
CompoundHydrogen iodideFormulaHIAppearancecolourless gasUse or occurrencestrong mineral acid
Is i2 soluble in water?
Nearly insoluble in water but very soluble in aqueous solutions of iodides.
Is copper iodide a precipitate?
We’ve already seen that copper(I) iodide is produced as an off-white precipitate if you add potassium iodide solution to a solution containing copper(II) ions. The copper(I) iodide is virtually insoluble in water, and so the disproportionation reaction does not happen.
What is the name for cul?
COPPER (II) CHLORIDE.
How do you write iodide formula?
Chemical formulaI−Molecular weight126.90447 g/molConjugate AcidHydrogen iodideFunctions asan antioxidantStd molar entropy−7.2 °C
Is copper iodide a solid?
Copper(I) iodide is a white solid. It does not dissolve in water.



