What is a solution of water
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. … The word aqueous (which comes from aqua) means pertaining to, related to, similar to, or dissolved in, water.
What are the 5 examples of solution?
S.NoTypes of SolutionExamples2Solid-liquidThe solution of sugar, salt etc in water.3Solid-gasSublimation of substances like iodine, camphor etc into the air.4Liquid-solidHydrated salts, mercury in amalgamated zinc, etc.5Liquid-liquidAlcohol in water, benzene in toluene
Is water a solution and why?
A solution is a homogeneous MIXTURE of two or more substances. Water is a compound, a pure substance. It is made of then elements hydrogen and oxygen, but shares none of their properties. … Pure water is a solvent, not a solution.
What forms a solution with water?
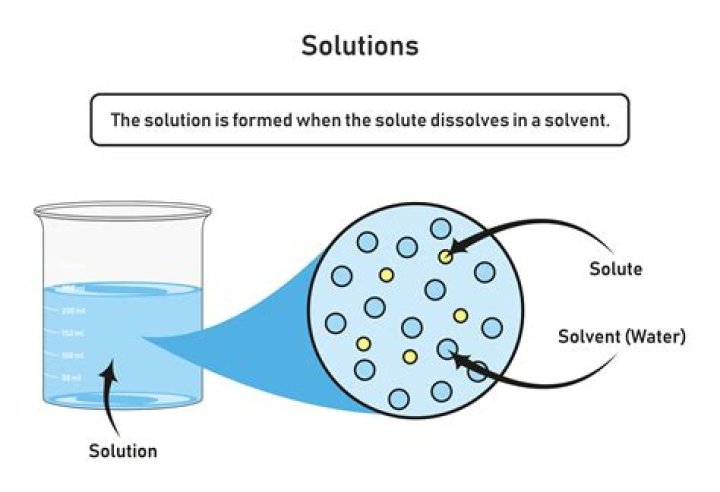
A solution is made when one substance called the solute “dissolves” into another substance called the solvent. … This break up is caused by coming into contact with the solvent. In the case of salt water, the water molecules break off salt molecules from the larger crystal lattice.What is solution short answer?
A solution is a homogeneous mixture of one or more solutes dissolved in a solvent. solvent: the substance in which a solute dissolves to produce a homogeneous mixture. solute: the substance that dissolves in a solvent to produce a homogeneous mixture.
Is water a solvent?
Water is called the “universal solvent” because it is capable of dissolving more substances than any other liquid. … It is water’s chemical composition and physical attributes that make it such an excellent solvent.
What are the examples of liquid solution?
SolutionSoluteExamplesliquidliquidalcoholic beverage (ethanol in water), gasolineliquidsolidtea, salt watersolidgasH2 in Pd (used for H2 storage)solidsolidmercury in silver or gold (amalgam often used in dentistry)
What type of mixture is a solution?
A solution is a mixture that is the same or uniform throughout. Think of the example of salt water. This is also called a “homogenous mixture.” A mixture that is not a solution is not uniform throughout.Is a solution always a liquid?
Answer: The solution is usually a liquid, but not always. It might also include a mixture of two solids like in alloys, and a mixture of gases, such as air.
What is solution and mixture?Mixtures are materials that contain two or more chemical substances dispersed among each other (mixed together). … Solutions are homogenous mixtures: particles of one substance (the solute) are mixed together with the particles of another substance (the solvent) – eg salty water.
Article first time published onIs water a solution or mixture?
Solvent PhaseSolute PhaseExamplesolidsolidsteel alloys
What is solution Class 9th?
A solution is a homogeneous mixture of two or more substances. … A solution consists of a solute and a solvent. The solute is the substance that is dissolved in the solvent. The amount of solute that can be dissolved in solvent is called its solubility.
What is solution and types of solution?
A solution is a homogeneous mixture of solvent and solute molecules. … A solution can be liquid, solid, or gaseous. Further, a solution can be a mixture of liquids, gases and solids. In some cases, like seawater, the solution consists of a great many different types of solutes, like salts, oxygen, and organic molecules.
Is milk a solution?
Well, from a general perspective, we would say milk is a solution because it is basically a mixture containing proteins (casein and whey), lactose, trace elements, lipids, fats and other substances in water without any bonding. … We classify it in this category because milk has more than one phase suspended in it.
Is ice and water a solution?
Both ice and water are chemically the same. Since solution is formed by chemically different substances, therefore, ice in water is not a solution.
Can a solution be two liquids?
Or perhaps you thought of a solid like salt or sugar that is completely dissolved in a liquid like water. This is a great example, but the world of solutions is actually much more impressive. A solution can also be made of two or more liquids, two or more gases, liquids and gases, and theoretically even two solids.
Is shampoo a liquid liquid solution?
Shampoo is more appropriately described as a colloid with the continuous phase being the liquid and the dispersed phase being a combination of oils and solids. As Rachna Rastogi mentions, it is a mixture of surfactants and water where the surfactants help suspend the solids in the water.
Which type of solvent is water?
Water is a protic solvent. Aprotic solvents such as acetone or dichloromethane tend to have large dipole moments (separation of partial positive and partial negative charges within the same molecule) and solvate positively charged species via their negative dipole.
What is water explain?
water, a substance composed of the chemical elements hydrogen and oxygen and existing in gaseous, liquid, and solid states. It is one of the most plentiful and essential of compounds. A tasteless and odourless liquid at room temperature, it has the important ability to dissolve many other substances.
When the solvent is water the solution is called?
An aqueous solution is a solution in which water is the solvent.
Why solution is a liquid?
The average intermolecular forces in the solution is stronger than in the individual liquids.
Is the air a solution?
Air is a solution made up of many gases. … There is more nitrogen than any other gas in air, so it is considered the solvent in an air solution.
What is a solid solution example?
A mixture of elements at the atomic level. Metals used in dentistry which readily form solid solutions with gold are copper, platinum, palladium, and silver. Steel is an example of a solid solution of a small amount of carbon in iron.
What is homogenous and heterogenous mixture?
A mixture is composed of one or more pure substances in varying composition. … Heterogeneous mixtures have visually distinguishable components, while homogeneous mixtures appear uniform throughout. The most common type of homogenous mixture is a solution, which can be a solid, liquid, or gas.
What are the 4 types of solutions?
S. no.SoluteSolution is called as1.GasFoam2.LiquidEmulsion3.LiquidGel4.SolidSolid Sol
Is water and flour a solution?
When you add flour to water, the mixture turns cloudy, and you cannot see through it. This mixture is not a solution but a suspension. In a the particles are larger than those found in a solution. Instead of dissolving, these larger particles turn the liquid cloudy.
Is salt water a solution?
Salt water is a solution because it has these two characteristics: it has the same concentration of each of its parts throughout the solution, and it can be separated by some physical process. … Water cannot be separated by any physical means. It would take a chemical change to separate water back into its parts.
What is a solvent in chemistry?
solvent, substance, ordinarily a liquid, in which other materials dissolve to form a solution. Polar solvents (e.g., water) favour formation of ions; nonpolar ones (e.g., hydrocarbons) do not.
What is a solution process?
The formation of a solution from a solute and a solvent is a physical process, not a chemical one. Substances that are miscible, such as gases, form a single phase in all proportions when mixed. Substances that form separate phases are immiscible.
Is water is a mixture?
Therefore, water is not a mixture; it is a compound and it is pure.
What is a solution in science?
solution, in chemistry, a homogenous mixture of two or more substances in relative amounts that can be varied continuously up to what is called the limit of solubility. The term solution is commonly applied to the liquid state of matter, but solutions of gases and solids are possible.